How is vacuum actually generated? – Part 3
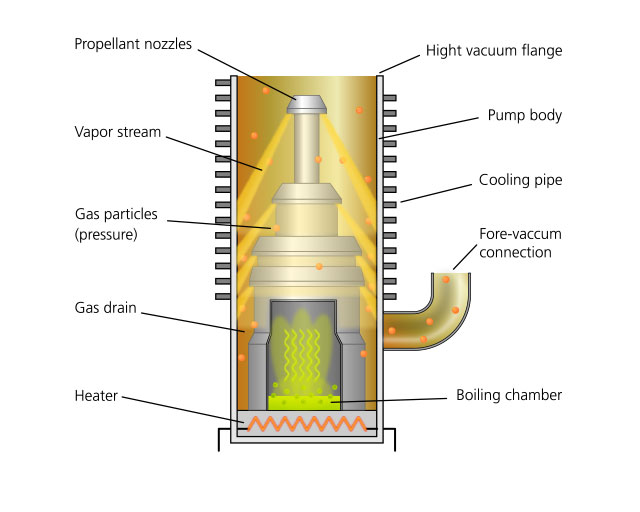
The high vacuum range with pressures smaller than 10-3 mbar is the domain of molecular flow. As a result of the high mean free path lengths in the range from centimeters to decimeters, interaction between the remaining gas particles barely occurs. Pumps for the high-vacuum range act as “one-way streets” for the highly diluted gas by administering impulses to individual particles in the conveying direction. The principle of the propellant pump was presented in the previous article. With the oil diffusion pump, oil vapor exits nozzles (see adjacent figure) at high speed into the suction chamber, compresses the residual gas and conveys it to the fore-vacuum connection. Oil diffusion pumps have no moving components; in industrial vacuum technology, they are therefore viewed as robust and virtually indestructible workhorses. The largest of them achieve a pumping speed of up to 50,000 l/s; final pressures of up to 10-8 mbar can be attained. At a maximum compression ratio of 106, however, compression to atmospheric pressure is not possible, as a result of which fore-vacuum connection must be installed upstream.
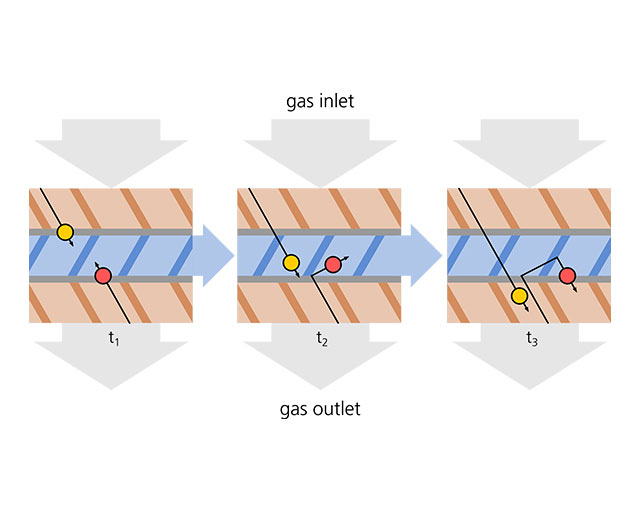
The turbomolecular pump offers an oil-free vacuum; moving surfaces serve as “propellants” here. The rotor of a turbomolecular pump is shaped like a turbine with a package of discs with oblique slits. It rotates at high speed within a package of stator discs which have slits in the opposite direction. The pump consists of 10 to 12 rotor-stator disc pairs. The pump mechanism is illustrated in the adjacent figure. Gas particles pass through a labyrinth from which there is no return.
The speed of the rotor must be adapted to the thermal speed of the species to be pumped. At the outer circumference of the rotor, the effective velocity is in the order of the speed of sound. Depending on their construction size, turbomolecular pumps achieve pumping speeds of between 50 l/s and 3,000 l/s which, however, drop sharply between 10-3 and 10-2 mbar, particularly in the case of light gases such as H2 or He.
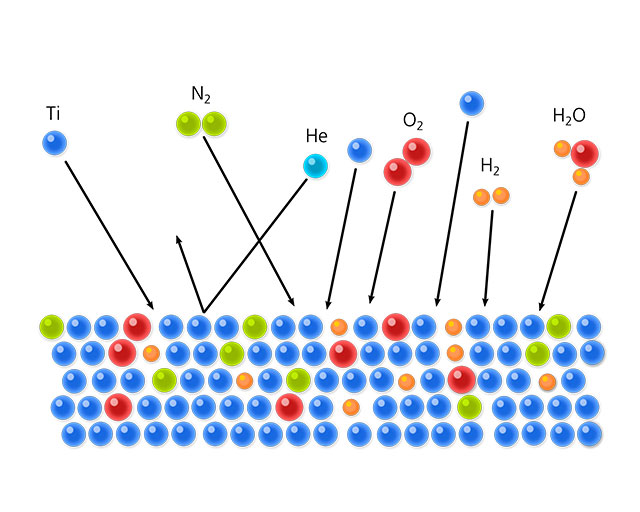
Getter and cryopumps reduce the achievable final pressure far into the ultra-high vacuum, namely to around 10-12 mbar. Whilst such pressures are no longer relevant in industrial coating facilities, for the large particle accelerators, which are utilized in order to accelerate electrons or ions up to the speed of light, they are important. (At 10-12 mbar, the mean free path length is around 50,000 kilometers!) With the getter pump, residual gas is stored in metallic surfaces as shown in the adjacent figure. Titanium is the preferred getter material. Getter surfaces are parts of the inner surfaces of the vacuum chamber. In order to maintain the pumping effect, the active layer must be constantly renewed. This is performed through evaporation or atomization of an appropriate material source.
Condensation on extremely cold surfaces is a further method for the removal of residual gas from a container. We are familiar with such phenomena in daily life: The bathroom mirror “pumps” water vapor when it steams up, just as the windscreen of a car parked on the road during a cold, damp winter night “pumps” water vapor, as a result of which it freezes. Cryopumps work with closed helium circuits. At the temperature of the liquid helium of 4.2 K, all gases condense on corresponding cold surfaces. The ice layer on a windshield has, thank goodness, a finite thickness, as the pumping effect diminishes with the ice formation. Cryopumps must also be regularly regenerated, i.e. baked out. This makes their application in continuously utilized vacuum systems somewhat laborious.
The pumping speed of getter pumps lies between 103 and 104 l/s, whilst that of cryopumps is between 102 and 105 l/s. Both pump types can, however, only be utilized at pressures below 10-4 mbar.
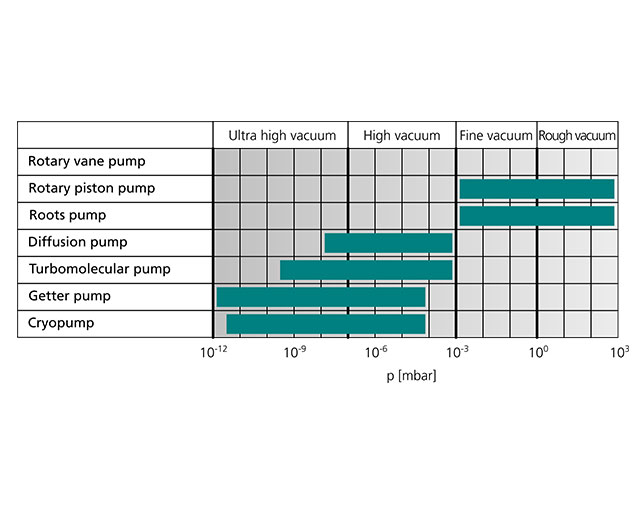
To conclude our excursion into the fundamentals of vacuum technology, the adjacent figure summarizes the most important pump types and their areas of application.
The role of the quantum leap for the formation of plasma will be clarified in the next article.
Last modified: